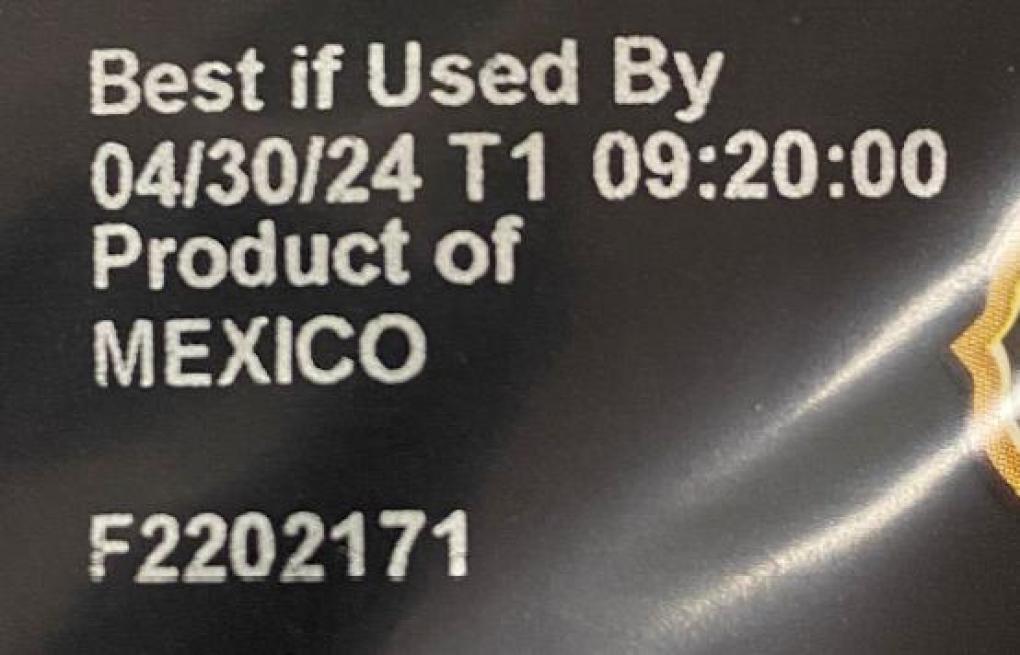Townsend Farms Inc. of Fairview, Oregon out of an abundance of caution is voluntarily recalling specific frozen fruit products linked to mango chunks supplied by SunOpta (Sunrise Growers) due to potential Listeria monocytogenes contamination.
Listeria monocytogenes is an organism which can cause serious and sometimes fatal infections in young children, frail or elderly people, and others with weakened immune systems. Although healthy individuals may suffer only short-term symptoms such as high fever, severe headache, stiffness, nausea, abdominal pain and diarrhea, Listeria infection can cause miscarriages and stillbirths among pregnant women. Any consumers concerned about an illness should contact their physician.
The product was distributed to Kroger Stores in the following states: Alabama, Arkansas, Georgia, Illinois, Indiana, Kentucky, Louisiana, Michigan, Missouri, Mississippi, Ohio, South Carolina, Tennessee, Texas, Virginia, and West Virginia; and under the following banners: Baker’s, Dillons, Food 4 Less, FoodsCo, Fry’s, Gerbes, Jay C, Mariano’s, Metro Market, Pay Less, Pick n’ Save, Ralphs, Ruler, and Smith’s from November 06, 2022 to March 16, 2023.
This voluntary recall includes the following products date codes. Date codes can be found printed on the back panel of the packaging.
UPC 0 11110 84663 1 Private Selection 48oz Tropical Mango Chunks
Date Codes:
BEST IF USED BY 04/30/24 T1 PRODUCT OF MEXICO F2202171
BEST IF USED BY 04/30/24 T2 PRODUCT OF MEXICO F2202172
BEST IF USED BY 04/30/24 T1 PRODUCT OF MEXICO F2202182
BEST IF USED BY 04/30/24 T2 PRODUCT OF MEXICO F2202183
BEST IF USED BY 05/04/24 T1 PRODUCT OF MEXICO F2202225
BEST IF USED BY 05/04/24 T1 PRODUCT OF MEXICO F2202229
BEST IF USED BY 05/07/24 T4 PRODUCT OF MEXICO F2202236
BEST IF USED BY 05/07/24 T4 PRODUCT OF MEXICO F2202241
BEST IF USED BY 07/18/24 T3 PRODUCT OF MEXICO F2300116
UPC 0 11110 87853 3 Private Selection 16oz Strawberry, Mango & Pineapple Blend
Date Codes:
BEST IF USED 04/30/24 T4 PRODUCT OF USA, MEXICO & COSTA RICA F2202174
BEST IF USED 04/30/24 T4 PRODUCT OF USA, MEXICO & COSTA RICA F2202185
BEST IF USED 05/04/24 T2 PRODUCT OF USA, COSTARICA & MEXICO F2202227
BEST IF USED 05/28/24 T2 PRODUCT OF PERU, MEXICO & COSTA RICA F2202332
UPC 0 11110 82026 6 Private Selection 48oz Classic Fruit Medley
Date Codes:
BEST IF USED 05/03/24 T3 PRODUCT OF USA, MEXICO, CHILE & COSTARICA F2202213
BEST IF USED 05/03/24 T3 PRODUCT OF USA, MEXICO, CHILE & COSTA RICA F2202220
BEST IF USED 05/04/24 T3 PRODUCT OF USA, MEXICO, CHILE & COSTA RICA F2202226
BEST IF USED 05/04/24 T3 PRODUCT OF USA, MEXICO, CHILE & COSTA RICA F2202230
BEST IF USED 05/10/24 T3 PRODUCT OF USA, MEXICO, PERU, CHILE & COSTA RICA F2202268
BEST IF USED 05/10/24 T3 PRODUCT OF USA, MEXICO, PERU, CHILE & COSTA RICA F2202273
BEST IF USED 05/18/24 T3 PRODUCT OF USA, MEXICO, CHILE, PERU & COSTA RICA F2202319
BEST IF USED 05/18/24 T3 PRODUCT OF USA, MEXICO, CHILE, PERU & COSTA RICA F2202325
UPC 0 11110 89623 0 Private Selection 48oz Blueberries, Strawberries & Mangoes
Date Codes:
BEST IF USED 04/30/24 T3 PRODUCT OF USA & MEXICO F2202173
BEST IF USED 04/30/24 T3 PRODUCT OF USA & MEXICO F2202184
BEST IF USED 05/07/24 T3 PRODUCT OF USA & MEXICO F2202235
BEST IF USED 05/07/24 T3 PRODUCT OF USA & MEXICO F2202240
BEST IF USED 05/08/24 T6 PRODUCT OF USA & MEXICO F2202247
The affected retail customers have been notified of this recall and instructed to remove any recalled product from retail store shelves and inventories.
Customers are urged to check their freezers for the recalled product, not to consume it and either discard the product or return it to the store for a full refund. Consumers may also request a refund by calling Kroger Customer Connect at 800-632-6900, Option 1, Monday through Friday, 7 a.m.- Midnight, EST. Saturday-Sunday, 7 a.m.-9:30 p.m., EST.
Consumers seeking product information may email recalls@townsendfarms.com or call 503-512-1317. Monday through Friday, 7 a.m.-Midnight, EST; Saturday-Sunday, 7 a.m.-9:30 p.m., EST.
To date, there have been no illnesses associated with this voluntary recall.
This voluntary recall is being conducted with the knowledge of the U.S. Food and Drug Administration (FDA).


Bulloch Public Safety
06/05/2026 Booking Report for Bulloch County
Chattooga Local News
WALKER COUNTY ENACTS SPAY / NEUTER ORDINANCE

Chattooga Schools
Forbes Gives Berry College A+ For Financial Health

Bulloch Public Safety
06/04/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/11/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/18/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/07/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/08/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/14/2026 Booking Report for Bulloch County