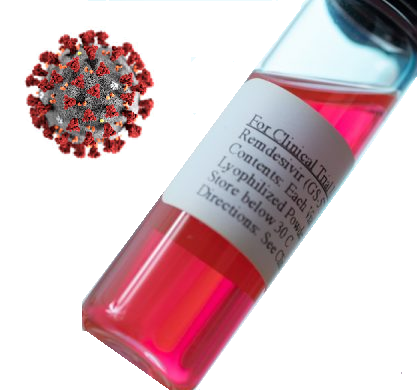
This weekend, the Georgia Department of Public Health (DPH) is distributing an initial allotment of the drug remdesivir received from the federal government. Georgia received 30 cases, with 40 vials of the drug per case, enough to treat about 110 patients, depending on the duration of an individual’s treatment.
Remdesivir is an antiviral medicine being used to treat hospitalized patients with serious symptoms caused by COVID-19 like low oxygen levels or pneumonia. It has been found to shorten the duration of disease in patients being treated in inpatient hospital settings. Remdesivir is given intravenously (IV) and decreases the amount of coronavirus in the body, helping patients recover faster.
The distribution plan for remdesivir in Georgia was developed by DPH leadership, including district health directors and emergency preparedness staff, in accordance with Food and Drug Administration (FDA) guidelines for its use. It is based on the number of patients on ventilators, the most severely ill, and clinical best practices.
Georgia hospitals receiving remdesivir reported 10 or more COVID-19 positive patients on ventilators, in addition to patients currently being treated with extracorporeal membrane oxygenation (ECMO), a machine that takes over the work of the heart and lungs. These criteria are subject to change based on the availability of remdesivir and the development of patient care at hospital facilities across the state.
The following hospitals are receiving remdesivir; Tift Regional Medical Center, Northeast Georgia Medical Center, Wellstar Kennestone Hospital, Phoebe Putney Memorial Hospital, Grady Health System, Emory University Hospital, Emory University Hospital Midtown, and Augusta University Medical Center.
“DPH is pleased to have the opportunity to share this promising treatment with hospitals on the front lines in the fight against COVID-19,” said Kathleen E. Toomey, M.D., M.P.H., DPH commissioner. “While this drug is not a cure for COVID-19, getting it into the hospitals and improving patient outcomes is moving in the right direction.”
Georgia has received a second, much larger allotment of remdesivir. DPH is surveying hospitals statewide over the weekend to determine need. This second allotment will be distributed next week.
Gilead Sciences, Inc. committed to supplying approximately 607,000 vials of the experimental drug over the next six weeks to treat an estimated 78,000 hospitalized COVID-19 patients under an emergency use agreement (EUA). The donation to the United States is part of 1.5 million vials of remdesivir the company is donating worldwide.
Remdesivir has not been approved by the FDA for widespread use because it is considered investigational and it is still being studied. Remdesivir was originally developed for use against Ebola. Clinical trials for remdesivir were done in Georgia at Emory University Hospital.
This is a press release from the Georgia Department of Public Health.

2 Comments
Leave a Reply
Cancel reply
Leave a Reply

Chattooga Lifestyle
Still Here: When Graduation Season Hurts After Child Loss

Bulloch Public Safety
05/22/2026 Booking Report for Bulloch County

Chattooga Schools
Trion High School Graduation Set for Tonight

Bulloch Public Safety
05/21/2026 Booking Report for Bulloch County

Bulloch Public Safety
04/27/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/04/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/11/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/07/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/18/2026 Booking Report for Bulloch County






NELDA SMITH
May 16, 2020 at 4:39 pm
WHY ? REMDESIVIR HAS SIDE EFFECTS AND TAKES 11 DAYS TO WORK AT $1000 A DOSE ! HYDROXYCHLOROQUININE HAS NEAR NO SIDE EFFECTS AND IMPROVEMENTS ARE SHOWN IN 5 HOURS AT 56 CENTS A DOSE. HYDROXYCHLOROQUININE TREATS / CURES THE COVID-19 “VIRUS” AND IT’S MUTATIONS. HYDROXYCHLOROQUININE IS ALSO A PREVENTATIVE (BETTER THAN A VACCINE WITHOUT VACCINE SIDE EFFECTS ). GEORGIA DPH SHOULD BE PUSHING HYDROXYCHLOROQUININE WHICH HAS PROVEN SAFE AND EFFECTIVE FOR BABIES, CHILDREN, ADULTS AND THE ELDERLY.
NELDA SMITH
May 16, 2020 at 4:46 pm
I AM HYPER-SENSITIVE TO MEDICATION. I WILL STICK TO MY 56 CENTS A PILL HCQ WITH NO SIDE-EFFECTS. REMDESIVIR HAS SERIOUS SIDE EFFECTS AND COST $1,000 A DOSE. WHY ON EARTH WOULD GA. DPH PUSH REMDESIVIR INSTEAD OF GIVING ALL GA. CITIZENS SAFE, EFFECTIVE, FAST ACTING AND INEXPENSIVE HYDROXYCHLOROQUININE ?