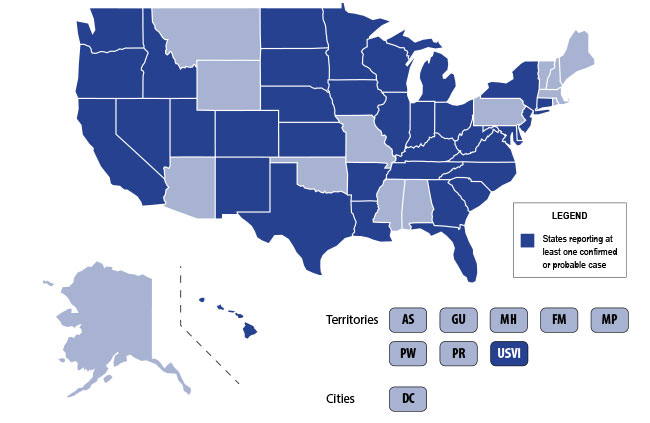
The CDC announced this week that both the numbers of confirmed cases of lung disease associated with e-cigarette product use maintained by the CDC and the states has increased.
Officials also said Thursday that the new case count is the first national aggregate based on the new CDC case definition developed and shared with states in late August.
Cases
- As of September 11, 2019, 380 confirmed and probable cases of lung disease associated with e-cigarette product use, or vaping, were reported by 36 states and the U.S. Virgin Islands.
- The previous case count released by CDC was higher because it reported possible* cases that were still under investigation by states. The current number includes only confirmed** and probable*** cases reported by states to CDC after classification.
- CDC is no longer reporting possible cases or cases under investigation and states have recently received the new CDC case definition to classify cases. The classification process requires medical record review and discussion with the treating healthcare providers. The current number is expected to increase as additional cases are classified.
- CDC will continue to report confirmed and probable cases as one number because the two definitions are very similar and this is the most accurate way to understand the number of people affected.
*A possible case is one still under investigation at the state level.
**A confirmed case is someone who recently used an e-cigarette product or vaped, developed a breathing illness, and for whom testing did not show an infection. Other common causes of illness have been ruled out as the primary cause.
***A probable case is someone who recently used an e-cigarette product or vaped, developed a breathing illness, and for whom some tests have been performed to rule out infection. Other common causes of illness have been ruled out as the primary cause.
Deaths
- Six total deaths have been confirmed in six states: California, Illinois, Indiana, Kansas, Minnesota, and Oregon.
The CDC is currently coordinating a multistate investigation and providing database information to the states as well as technical assistane, including working closely with affected states to characterize the exposures and the extent of the outbreak.
The organization is also working closely with the Food and Drug Administration (FDA) to collect information about recent e-cigarette product use, or vaping, among patients and to test the substances or chemicals within e-cigarette products used by case patients.
The FDA also announced this week that it intends to finalize an enforcement policy that will clear non-tobacco-flavored e-cigarettes from the market.


Bulloch Public Safety
05/29/2026 Booking Report for Bulloch County

Chattooga Lifestyle
Column: The Waiting List Has Names

Bulloch Public Safety
Man Arrested in Burke County for Statesboro Kidnapping and Arson

Bulloch Public Safety
05/28/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/04/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/11/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/18/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/07/2026 Booking Report for Bulloch County

Bulloch Public Safety
05/01/2026 Booking Report for Bulloch County